 .
.
Chemicals currently represented in the simulation include
Beta-amyloid protein, interleukins IL-1B, IL-6 and
tumor necrosis factor, TNF. (The former is responsible for initiating
an inflammatory response, the latter three
are cytokines secreted by glial cells.)
Chemicals are produced and taken up by cells, diffuse throughout
the domain and influence cell behaviour as described above in
The cells.
The diffusion coefficient of a substance is
computed from its molecular weight,
(Goodhill, 1998, 1997)
and corrected for the overall effect of
tortuosity of brain tissue
(Sykova, 1997;
Nicholson and Sykova 1998), and
for possible local effects
that affect diffusion (described below).
For S(x,y,t) the
concentration of some chemical at point (x,y) at time,
t, and given its diffusion coefficient at point (x,y),
i.e. D(x,y), the following partial differential equation
(PDE) describes diffusion of the chemical,
 .
.
Both explicit and implicit finite difference methods for numerically
solving the chemical transport PDE were tested in our simulations.
The constraints of running a Java-based simulation online
have meant that we are currently using an explicit method in order to
save on memory.
In explicit methods, spatial derivatives use data
from the preceding time step. The discretized
PDE then has the form
 .
. .
.
A draw-back is that the above method is numerically stable only
if the maximum diffusivity, D, is less than
DX*DY/(4*dt) (for the case where DX = DY).
Our current formulation sets
DX = DY = 10 and dt = 0.0125 so that the maximum
diffusivity is D = 2000 microns2 per minute.
This stability constraint was used in selecting an appropriate
micro time scale.
The discretized form of the PDE in an implicit method
typically has the form:
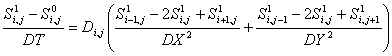 .
.The implicit method described above is the backwards Euler method. In the program, we use a generalized Crank-Nicholson scheme which averages the spatial derivatives between the old data and the new data. (The fact that one can weight the average is what makes it generalized.) In our current formulation, we invert an mn x mn matrix. This requires a huge computational overhead, but as long as the diffusion coefficients do not change, this only needs to be done once. If the diffusion coefficients change (as is currently the case), this method should be avoided. However, in such a case, another possibility is to use an Alternating Direction Implicit (ADI) method: this uses the fact that diffusion in each direction leads to a tridiagonal matrix which can be solved on its own, without building and inverting the entire mn x mn matrix. (There are also shortcuts for inverting a tridiagonal matrix which reduce the computational overhead.)
With an implicit method, one rarely has to worry about
stability. Thus, updates can take place on a
macro time scale. In practice, the consideration of numerical
accuracy generally restricts one to smaller time steps nevertheless.
Further, the savings due to fewer updates relative to the explicit schemes
may be outweighed by the additional computation power
needed to invert matrices. One must have the memory to store
the matrices, and
when multiple inversions are necessary, they carry a
cost of (mn)3 which can be enormous. It is
for these reasons that we currently use an explicit scheme.
Currently, the production of chemicals by cells is assumed to occur at a
constant rate, r. Thus, if a cell secretes some chemical,
S, into a grid space, the rate of change of S in that
grid space is given by the equation
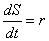 .
.Amyloid protein occurs in soluble and fibrous forms. In the soluble form, it is secreted by (live) neuronal tissue, diffuses in the tissue, interacts with amyloid fibers, and is removed by microglia.
A source of soluble amyloid secreted from
neurons in the center of the environment
forms the initial "stimulus" at the begining of a simulation.
Amyloid fibers are placed randomly in the domain with frequency
based on the initial fiber
occupancy parameter, p.
(Initially, for
each grid space,
a random variable uniformly distributed between 0 and 1
is generated; if this value is less than p, a random
concentration of fibers uniformly distributed between 0 and
MAXFIBERS is placed in the grid space.)
When a neuron's internal concentration of IL-1B exceeds
the source triggering level the Neuron
secretes soluble amyloid
protein at its grid location. The rate of
secretion, r, is determined as a function of the
source concentration parameter, I. Specifically,
Soluble amyloid diffuses according to the diffusion
properties discussed above. Initially, the diffusion coefficient
equals the diffusivity parameter throughout the region. However,
in regions where astrocytes are activatedm
astrocyte blocking may partially
seal off certain regions and thus reduce the
diffusion coefficient of chemicals in the given region.
Exchange between soluble and fibrous amyloid occurs in two ways. Fibers already present in any site can grow in the presence of soluble amyloid (sol to fiber transition rate parameter governs this rate). Fibers in the surrouding grid spaces can elongate into neighboring sites. In either case, the concentration of soluble amyloid in the grid space must exceed the critical sol-AB for fibers parameter, for fiber growth to occur.
When fibers are present, growth occurs at a rate proportional to
the product of the average surrounding fiber concentration, F
(a weighted average with the center fibers counting more than the
immediately surrounding fibers by a programmer defined constant,
WEIGHT -- currently, WEIGHT=2.0, meaning that fibers in
the center count twice as much as surrounding fibers), and the
difference between the concentration of soluble amyloid, s,
within the grid space and the critical sol-AB for fibers
parameter, b. If f is the concentration of fibers
in the grid space, then
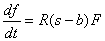
In the case where no fibers are present in the grid space under consideration, the grid space may still gain fibers if no astrocytes occupy the space and the concentration of soluble amyloid exceeds the critical sol-AB for fibers parameter. This is done through the process of fiber nucleation. Fiber nucleation can depend on (1) surrounding grid spaces having fibers or (2) purely on the amount of soluble amyloid.
In the first case, fiber nucleation depends on the weighted average of concentration of fibers surrounding the grid space, F, the maximum fiber concentration, MAXFIBERS, and the new fiber nucleation effectiveness parameter, n. These terms are combined to give a "probability" that the site undergoes nucleation, p = n*(F/MAXFIBERS). Every macro time step, nucleation is tested for via a Monte Carlo technique. Specifically, a uniformly distributed random number between 0 and 1 is generated. If its value is less than p, then nucleation occurs and the rate of fiber growth is determined as described in the preceding paragraph.
In the second case, fiber nucleation depends on the concentration
of soluble amyloid. The new fiber nucleation effectiveness
parameter, n, is divided by the maximum value that it
can attain from its slider, to determine a "probability" of nucleation,
p = n/max. Every macro time step,
DT, nucleation is
tested for via a Monte Carlo technique. Specifically, a uniformly
distributed random number between 0 and 1 is generated. If its
value is less than p, then nucleation occurs. In this case,
fiber growth is
IL-1B is secreted by microglia and
absorbed by astrocytes and
neurons. It is free to diffuse
throughout the environment.
Microglia secrete IL-1B into the
same grid space at which they are located when their internal
concentration of soluble amyloid exceeds the triggering
concentration parameter. The appropriate checks are made
so that the microglia concentration is taken into
account. The rate of secretion is the
product of the IL-1B secretion rate, u, and
the microglia concentration, q, so that
IL-1B diffuses according to the diffusion properties
discussed above. Initially, the diffusion coefficient
equals the diffusivity parameter throughout
space. However,
astrocyte blocking may reduce the
diffusion coefficient in specific regions.
IL-6 is secreted by astrocytes and
absorbed by neurons. It is free
to diffuse throughout the environment.
Astrocytes secrete IL-6 into the
same grid space at which they are located based on the amount
of IL-1B they have in storage, s. Adjusting for the
astrocyte concentration parameter, q, secretion
occurs as long as
s is greater than
IL-6 diffuses according to the diffusion properties
discussed above. Initially, the diffusion coefficient
equals the diffusivity parameter throughout
space. However,
astrocyte blocking may reduce the
diffusion coefficient in specific regions.
TNF is secreted by astrocytes and
absorbed by neurons. It is free
to diffuse throughout the environment.
Astrocytes secrete TNF into the
same grid space at which they are located based on the amount
of IL-1B they have in storage, s. Adjusting for the
astrocyte concentration parameter, q, secretion
occurs as long as s is greater than
TNF diffuses according to the diffusion properties
discussed above. Initially, the diffusion coefficient
equals the diffusivity parameter throughout
space. However,
astrocyte blocking may reduce the
diffusion coefficient in specific regions.